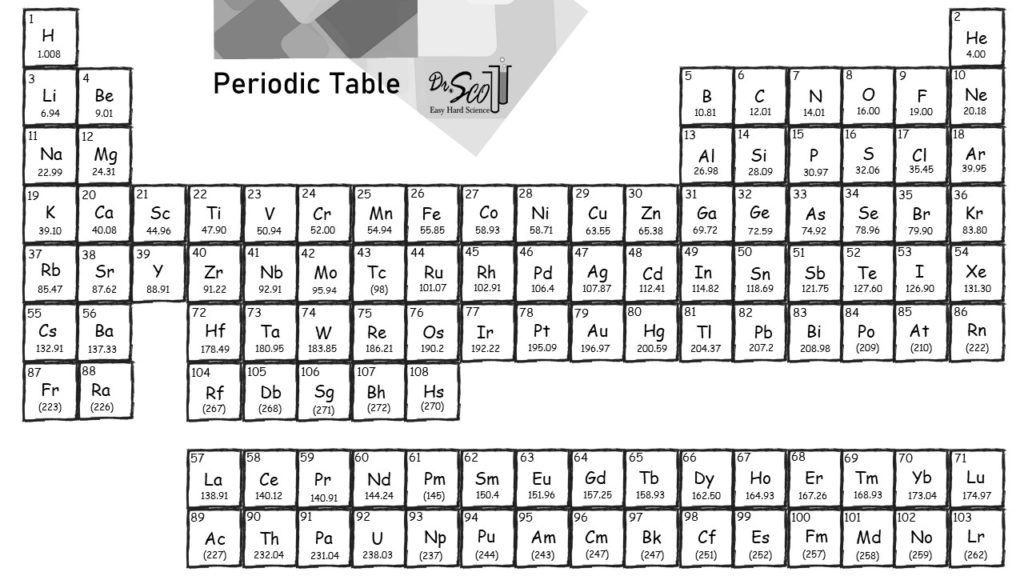
CO 2 has one carbon atom and two oxygen atoms.Helium is used as a cooling medium for the Large Hadron Collider (LHC), and the superconducting magnets in MRI scanners and NMR spectrometers. A colourless, odourless gas that is totally unreactive. Oxygen (O) has an atomic mass of about 16.00 amu. Helium was detected in the sun by its spectral lines many years before it was found on Earth.Carbon (C) has an atomic mass of about 12.01 amu.Let's calculate the molar mass of carbon dioxide (CO 2): Molar mass can be calculated by using the periodic table and following three simple steps. Add them together: add the results from step 3 to get the total molar mass of the compound.Calculate molar mass of each element: multiply the atomic mass of each element by the number of atoms of that element in the compound.The atomic mass is usually found on the periodic table and is given in atomic mass units (amu). Find atomic masses: look up the atomic masses of each element present in the compound.For example, water is H 2O, meaning it contains two hydrogen atoms and one oxygen atom. Identify the compound: write down the chemical formula of the compound.

One mole contains exactly 6.022 ×10 23 particles (Avogadro's number) Mole is a standard scientific unit for measuring large quantities of very small entities such as atoms and molecules.Use the buttons above to change your view of the periodic table and view Murray Robertson’s stunning Visual Elements artwork. Click the tabs at the top to explore each section. Visualize trends, 3D orbitals, isotopes, and mix compounds. The Royal Society of Chemistrys interactive periodic table features history, alchemy, podcasts, videos, and data trends across the periodic table. Molar mass ( molar weight) is the mass of one mole of a substance and is expressed in g/mol. Interactive periodic table showing names, electrons, and oxidation states.(1 u is equal to 1/12 the mass of one atom of carbon-12) Converting the mass, in grams, of a substance to moles requires a conversion factor of (one mole of substance/molar mass of. If the mass of a substance is known, the number of moles in the substance can be calculated.
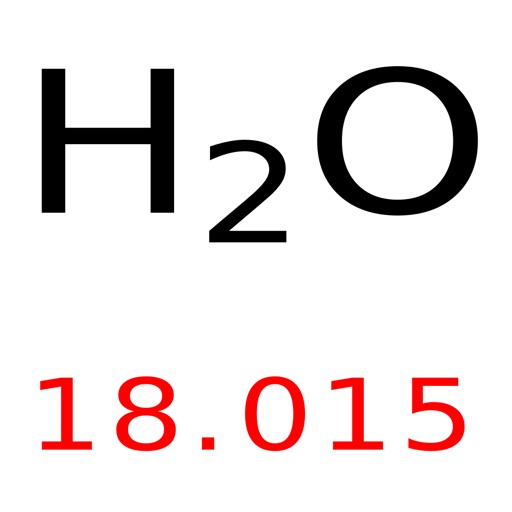
Molecular mass ( molecular weight) is the mass of one molecule of a substance and is expressed in the unified atomic mass units (u). The molar mass of an element is found on the periodic table, and it is the elements atomic weight in grams/mole (g/mol).To calculate molecular weight of a chemical compound enter it's formula, specify its isotope mass number after each element in square brackets.Įxamples of molecular weight computations: Molar mass calculator also displays common compound name, Hill formula, elemental composition, mass percent composition, atomic percent compositions and allows to convert from weight to number of moles and vice versa.Ĭomputing molecular weight (molecular mass) Functional groups: D, Ph, Me, Et, Bu, AcAc, For, Tos, Bz, TMS, tBu, Bzl, Bn, DmgĮxamples of molar mass computations: NaCl, Ca(OH)2, K4, CuSO4*5H2O, nitric acid, potassium permanganate, ethanol, fructose, caffeine, water.Element 43 (technetium), element 61 (promethium), and most of the elements with atomic number 84 (polonium) and higher have their atomic mass given in.
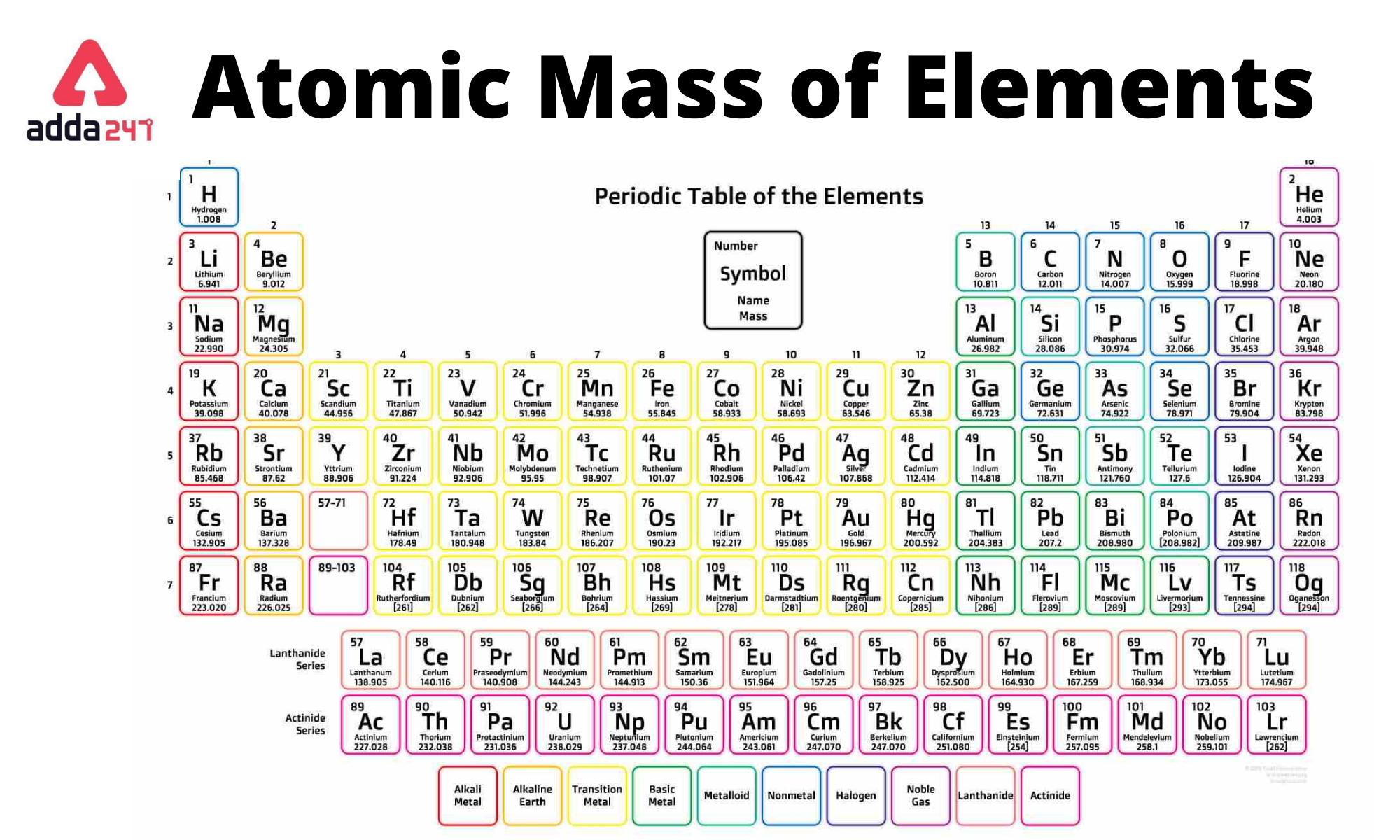
In studying the periodic table, you might have noticed something about the atomic masses of some of the elements. Capitalize the first letter in chemical symbol and use lower case for the remaining letters: Ca, Fe, Mg, Mn, S, O, H, C, N, Na, K, Cl, Al. Answer: (a) noble gas, (b) chalcogen, (c) alkaline earth metal, (d) alkali metal. The molar mass of a compound is the mass in grams of 1 mole of that substance, a property expressed in units of grams per mole (g/mol)īecause the definitions of both the mole and the formula mass are based on the same reference substance, 12C, the molar mass of any compound is numerically equivalent to its formula weight in amu.Chlorine gas appears as a pale yellow-green gas with a strong, pungent odor.Ĭomputing molar mass (molar weight)To calculate molar mass of a chemical compound enter its formula and click 'Compute'. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. Element Mercury (Hg), Group 12, Atomic Number 80, d-block, Mass 200.592. The masses of 1 mole of different compunds, however, are different, since the masses of the individual molecules are drastically different. Created by video journalist Brady Haran working with chemists at The University of Nottingham. Similar to atoms, 1 mole of any compound contains the same number of molecules as 1 mole of any other compund.
